|
When ClF3 comes into touch with any element, it turns into a poisonous gas that burns at around 2400 degrees Celsius. What Makes Chlorine Trifluoride So Dangerous? ClF3 also burns water, oxidizing it to produce oxygen, oxygen difluoride (OF2), hydrogen fluoride, and hydrogen chloride in controlled amounts: Many metals react to form chlorides and fluorides phosphorus reacts to form phosphorus trichloride (PCl3) and phosphorus pentafluoride (PF5), while sulfur reacts to form sulfur dichloride (SCl2) and sulfur tetrafluoride (SF4). It is made by fluorinating chlorine, which also produces ClF, and then distilling the mixture to separate it. The chemical irritates mucous membranes, eyes, and skin, and it can cause lung harm when inhaled as Chlorine Trifluoride gas. 
Vessels constructed of steel, copper, or nickel are not burned by ClF3 because a thin passivation layer of insoluble metal fluoride forms, preventing further corrosion nevertheless, molybdenum, tungsten, and titanium create volatile fluorides and are thus inappropriate.Ĭhloride Trifluoride is available in a condensed form on the market, and it changes into a pale-greenish yellow liquid when compressed at normal temperature. These are frequently aggressive and, in some cases, cause explosive reactions. It has high reactivity with most inorganic and organic materials, and it can start the combustion of many materials that are typically non-flammable without the use of an ignition source. Ruff and Krug were the first to report it in 1930 when they fluorinated chlorine to make ClF, which was then separated by distillation.ĬlF3 is an interhalogen compound with a high oxidizing and fluorinating ability. some fundamental understanding regarding the molecular geometry, usage, and applications of ClF3 is also discussed here in this article.Ĭhlorine trifluoride (ClF3) is a colorless, corrosive, and toxic gas utilized in plasmaless cleaning and etching procedures in the semiconductor industry, as well as rocket fuels, nuclear reactor fuel processing, and other industrial processes. Well, it is difficult to say a priori, will the bigger lp-bp repulsion outweigh the smaller lp-lp repulsion, but it assumed in VSEPR theory that it will, so the first structure is more favorable.The polarity of ClF3 will be discussed in this article based on polarity determining factors including its physical and chemical features. So, in the second structure the lp-lp repulsion is smaller, but the overall lp-bp repulsion is bigger while the overall bp-bp repulsion is basically the same. Six lp-bp repulsions at 90° (three bonding pairs out of plane interacts with two lone pairs). 
Two bp-bp repulsions at 90° and one at 180°.Four lp-bp repulsions at 90° (two bonding pairs out of plane interacts with two lone pairs).Two lp-bp repulsions at 120° (one bonding pair in the same plane interacts with two lone pairs).Yes, generally, lone pair - lone pair (lp-lp) repulsions are greater than lone pair - bonding pair (lp-bp) and bonding pair - bonding pair (bp-bp) ones, but the later could not be just neglected. OP is right in that lone pairs repulsion is smaller in the second structure, however, we have to consider repulsions between lone pairs and bonding pairs as well. You cannot put 4 electrons in a p-orbital. 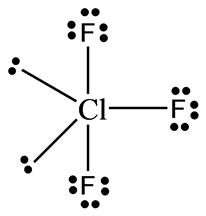
The structure on the left is "preferred" because the structure on the right cannot exist.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
